 NK cells are able to bind to and directly kill tumor cells. Avelumab is unique among approved ICIs because it is also able to enhance NK cell-mediated ADCC. Immunosuppressive signals in the TME are decreased via blockade of PD-1/PD-L1 interaction leading to the reinvigoration of effector T cells and antitumor activity. 
It can inhibit the interaction between PD-L1 and PD-1 and mediate ADCC through retention of its native FcR. Avelumab is an IgG1 monoclonal antibody with a dual mechanism of action. 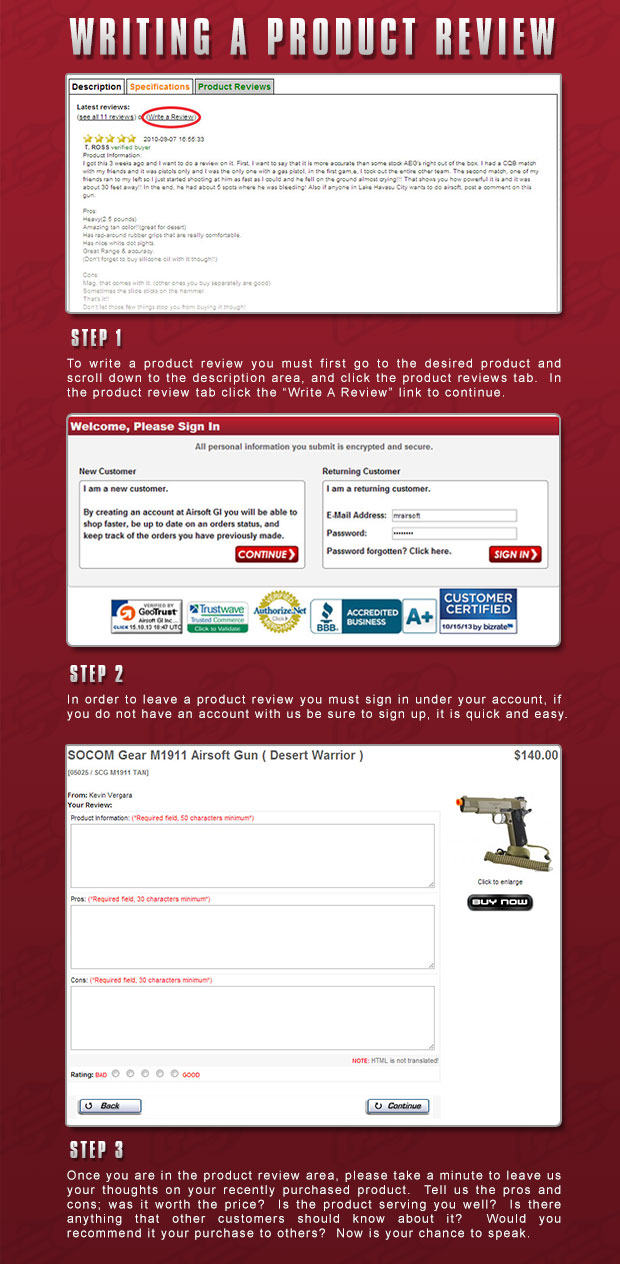
MSB0010718C Merkel cell carcinoma anti-PD-L1 avelumab immune checkpoint inhibitor immunotherapy monoclonal antibody urothelial carcinoma.Īvelumab’s dual mechanism of action. Its acceptable safety profile and ability to induce durable responses in otherwise deadly tumors provide the rationale for its use in other tumor types and in combination with other therapies. 
FDA for the treatment of metastatic Merkel cell carcinoma and metastatic urothelial carcinoma that has progressed during or following treatment with a platinum-based regimen. Most early clinical trials of avelumab as monotherapy and in combination regimens were part of the international JAVELIN clinical trial program, which included more than 7000 patients in more than 30 trials with at least 15 tumor types. Avelumab is a fully human monoclonal antibody that blocks the interaction between PD-L1 on tumor cells and PD-1 on T cells, thereby inhibiting immunosuppression in the tumor microenvironment and reducing tumor growth. Although immunotherapies have been employed for many decades, immune checkpoint inhibitors have only recently entered the oncologic landscape. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
